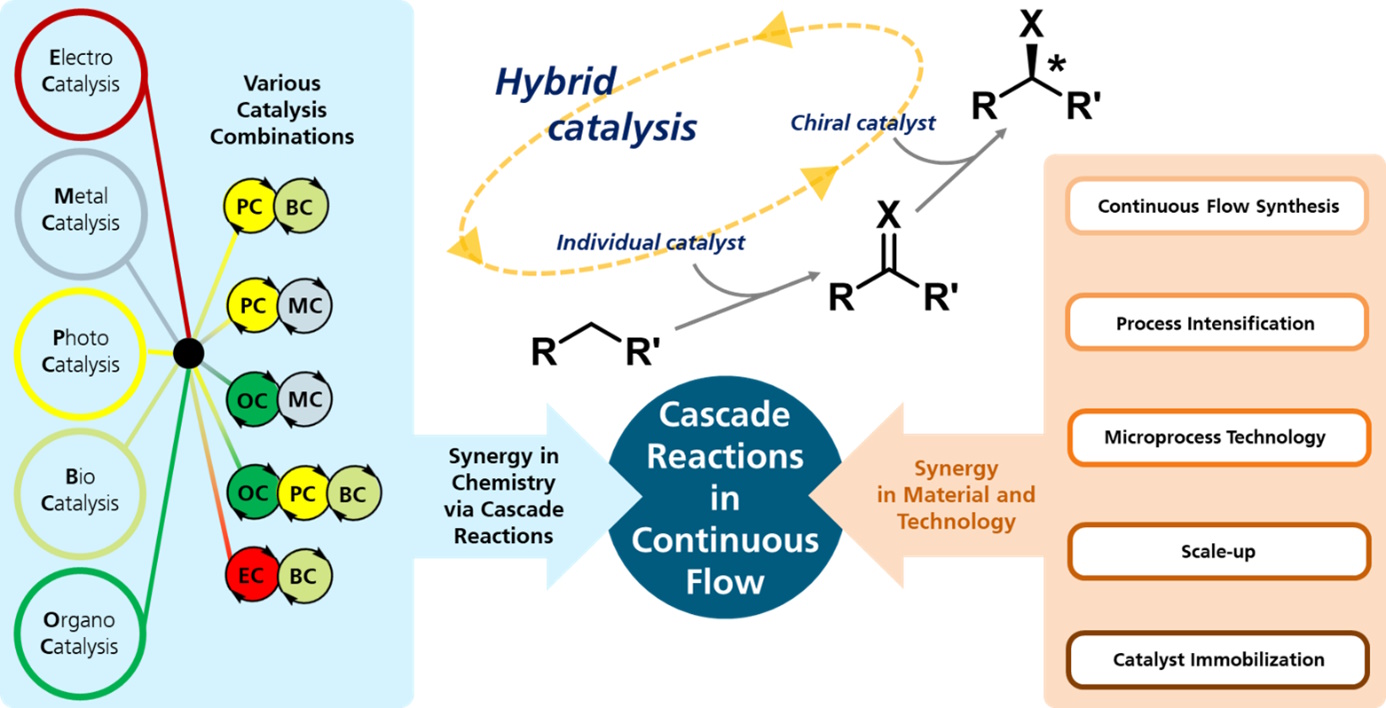
Are you looking for solutions for complex chemical processes?

Advancing Catalysis and Continuous Process Design
This concise summary offers insights into our latest developments in catalyst synthesis, reactor design, and the implementation of continuous cascade processes – key technologies for efficient and scalable chemical production.
Drawing on our multidisciplinary expertise, we support partners from concept to pilot scale with customized solutions tailored to their specific process requirements.
Download the full PDF to learn more – and contact us directly to explore how we can help bring your process ideas to life.
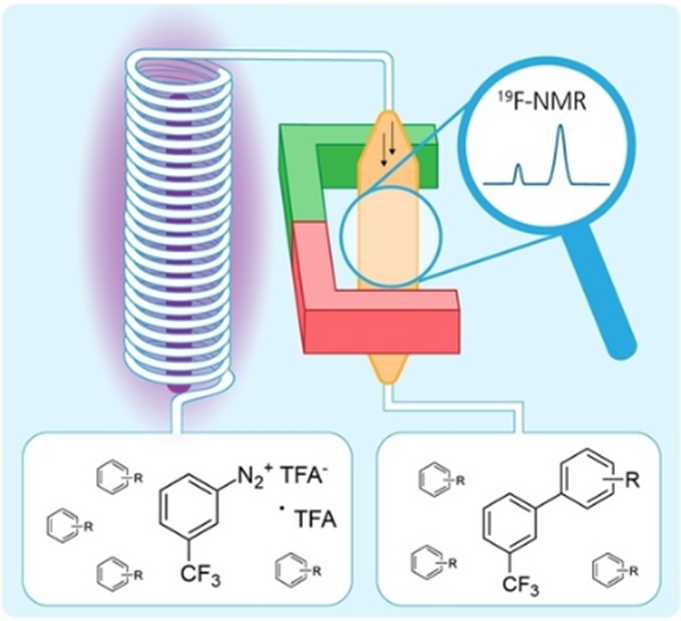
Cascade reactions for the continuous flow synthesis of fine chemicals